Welcome to Changzhou Unique Medical Co.,Ltd!
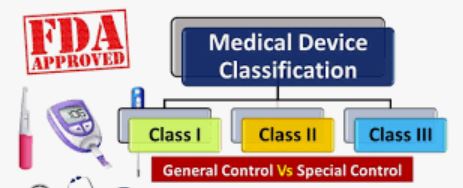
Medical devices class III are high risk to the patients and user. It requires special labelling requirement, guidelines, etc. These devices usually sustain or support life, are implanted, or present potential unreasonable risk of illness or injury. They represent 10% of commercially medical devices regulated by the FDA. Examples of Class III devices include implantable pacemakers and breast implants.